Importation of US-Authorized Vasopressin Injection, USP, 20 units/mL (For IV Infusion) Due to the Current Shortage of Canadian-Authorized Vasopressin Injection, USP, 20 units/mL (For IM or SC Administration)
Summary
See Key Messages below
Affected products
|
Product |
Dosage Form, Strength, and Packaging Format |
Country of |
Manufacturer |
Importer in Canada |
|---|---|---|---|---|
|
Vasopressin
|
Sterile, aqueous Preservative-free.
|
United States Product Code: Each vial: NDC 63323-930-00 1 mL Single- Unit of Sale: NDC 63323- A tray of 25 |
Fresenius Kabi
|
Fresenius Kabi
|
Issue
There is a shortage of Vasopressin Injection, USP, 20 units/mL (for intramuscular [IM] or subcutaneous [SC] administration) in Canada. This shortage presents serious risks to patients who may require vasopressin therapy in critical care settings, based on the clinical judgment of qualified healthcare professionals. Given the medical need, Health Canada has permitted the exceptional, temporary importation and sale of a similar but not identical product, US-authorized Vasopressin, USP, 20 units/mL (for intravenous [IV] infusion), with English-only labels by Fresenius Kabi USA, LLC.
Audience
Healthcare professionals including emergency medicine physicians, critical care physicians, anesthesiologists, nurse practitioners, critical care nurses, and pharmacists.
Key messages
- There is a shortage of Vasopressin Injection, USP, 20 units/mL (for intramuscular [IM] or subcutaneous [SC] administration) (DIN 02139502) in Canada. Given the serious risks this shortage presents to patients in critical care settings, Health Canada has permitted the exceptional, temporary importation and sale of a similar but not identical product, US-authorized Vasopressin Injection, USP, 20 units/mL (for intravenous [IV] infusion) with English-only labels by Fresenius Kabi USA, LLC.
- Healthcare professionals are advised to:
- Be aware that US-authorized Vasopressin Injection, USP (for IV infusion) differs significantly from the Canadian-authorized Vasopressin Injection, USP (for IM or SC administration) in indication, route of administration, packaging and formulation, dilution requirements, storage conditions and in-use periods, and safety information (see the Information for healthcare professionals section).
- Be aware that the US-authorized product is intended exclusively for IV infusion after dilution. It is not labelled for IM or SC use, and the safety of IM or SC administration has not been established.
- Consult the US Prescribing Information (USPI), available in English and French on the Fresenius Kabi Canada Ltd. website, as well as the US-authorized inner and outer labels (see Appendix 1), for complete instructions on the proper use of US-authorized Vasopressin Injection, USP (for IV infusion), including the indication, route of administration, dilution, dosing, storage, contraindications, warnings and precautions, and other important information.
Background
In Canada, Vasopressin Injection, USP, 20 units/mL, is authorized for IM or SC administration for the prevention and treatment of postoperative abdominal distention, in abdominal roentgenography to dispel interfering gas shadows, and in diabetes insipidus. Health Canada is aware that the Canadian-authorized product has also been used off-label to treat vasodilatory shock.
In the US, Vasopressin Injection, USP, 20 units/mL (for IV infusion) is indicated to increase blood pressure in adults with vasodilatory shock who remain hypotensive despite fluids and catecholamines.
Information for healthcare professionals
The US-authorized product has the same active ingredient (vasopressin) and strength (20 units/mL) as Fresenius Kabi Canada Ltd.’s Canadian-authorized Vasopressin Injection, USP (DIN 02139502). However, there are key differences in indication, route of administration, packaging and formulation, dilution requirements, and storage conditions and in-use periods, which are important to note (see Table 1). The Canadian- and US-authorized products also have differences in safety information, including contraindications, serious warnings and precautions, adverse reactions, and drug interactions, which are detailed in their respective prescribing information.
Table 1: Key differences between US-authorized Vasopressin Injection, USP, 20 units/mL (for IV infusion) and Canadian-authorized Vasopressin Injection, USP, 20 units/mL (for IM or SC administration).
| US-Authorized Vasopressin Injection, USP, 20 units/mL | Canadian-Authorized Vasopressin Injection, USP, 20 units/mL | ||
|---|---|---|---|
| Indications | Increase blood pressure in adults with vasodilatory shock who remain hypotensive despite fluids and catecholamines. |
|
|
| Route of Administration | IV infusion only. Must be diluted prior to infusion. | IM or SC | |
|
Single-dose vial. |
Multi-use vial. The stopper is not made with natural rubber latex. Each mL contains: 20 USP Vasopressin units; chlorobutanol (anhydrous) 5 mg as preservative; Water for Injection q.s.; Glacial acetic acid and/or sodium hydroxide for pH adjustment (2.5 - 4.5). |
|
| Dilution Requirements |
|
No dilution required for IM/SC administration. | |
|
Store unopened vials between 2°C and 8°C (may be used until manufacturer expiration date) or between 20°C to 25°C (may be used for 12 months or until manufacturer expiration date, whichever is earlier). Do not freeze. For diluted IV solution: discard unused diluted solution after 18 hours when stored at room temperature or 24 hours when refrigerated. |
Store between 15°C and 30°C; discard within 14 days after initial use (multi-use vial). Do not permit to freeze. |
Healthcare professionals are advised to:
- Be aware that US-authorized Vasopressin Injection, USP, 20 units/mL (for IV infusion) differs significantly from Canadian-authorized Vasopressin Injection, USP, 20 units/mL (for IM or SC administration) in indication, route of administration, packaging and formulation, dilution requirements, storage conditions and in-use periods, and safety information.
- The US-authorized product is intended exclusively for IV infusion after dilution. It is not labelled for IM or SC use, and the safety of IM or SC administration has not been established.
- Consult the US Prescribing Information (USPI), available in English and French on the Fresenius Kabi Canada Ltd. website, as well as the US-authorized inner and outer labels (see Appendix 1) for complete instructions on the proper use of US-authorized Vasopressin Injection, USP, 20 units/mL (for IV infusion), including the indication, route of administration, dilution, dosing, storage, contraindications, warnings and precautions, and other important information.
Images of US-authorized Vasopressin Injection, USP, which are in English-only and do not include French text, can be found in Appendix 1. Healthcare professionals are advised that the product labels and packaging may differ from the Canadian-authorized product. Proper selection of the intended product must be verified to avoid confusion with other products and prevent medication errors.
Additionally, the US-authorized product does not have a Drug Identification Number (DIN) or a barcode that scans in medication management systems in Canada. A facility-generated sticker is recommended to enable barcode scanning and allow proper identification of the product being dispensed and administered. It is the responsibility of the receiving organization to create this sticker.
Action taken by Health Canada
To help mitigate the shortage of Vasopressin Injection, USP in Canada, Health Canada has permitted the exceptional, temporary importation and sale of US-authorized Vasopressin Injection, USP by Fresenius Kabi USA, LLC. and added this product to the List of Drugs for Exceptional Importation and Sale.
Health Canada has worked with Fresenius Kabi Canada Ltd. to prepare this alert for Vasopressin Injection, USP. Health Canada is communicating this important safety information to healthcare professionals and Canadians via the Recalls and Safety Alerts Database on the Healthy Canadians Web Site. This communication update will be further distributed through the MedEffectTM e-Notice email notification system.
Report health or safety concerns
Managing health product-related side effects depends on healthcare professionals and consumers reporting them. Any case of serious or unexpected side effects in patients receiving Vasopressin Injection, USP should be reported to Fresenius Kabi Canada Ltd. or Health Canada.
Fresenius Kabi Canada Ltd.
165 Galaxy Blvd., Suite 100, Toronto, ON, M9W 0C8, Canada
Phone: 1-877-779-7760
Fax: 1-844-605-4465
E-mail: Canada_Vigilance@fresenius-kabi.com
For questions or concerns about US-authorized Vasopressin Injection, USP, 20 units/mL, please contact Fresenius Kabi Canada Ltd. at Canada_medinfo@fresenius-kabi.com OR by phone at 1-877-779-7760.
To correct your mailing address or fax number, contact Fresenius Kabi Canada Ltd.
You can report any suspected adverse reactions associated with the use of health products to Health Canada by:
- Calling toll-free at 1-866-234-2345; or
- Visiting MedEffect Canada's Web page on Adverse Reaction Reporting for information on how to report online, by mail or by fax.
For other health product inquiries related to this communication, contact Health Canada at:
Regulatory Operations and Enforcement Branch
E-mail: hpce-cpsal@hc-sc.gc.ca
Telephone: 1-800-267-9675
Original signed by
Anabela Costa
Vice President, Scientific Affairs
Appendix 1
A. Image of US-Authorized Vasopressin Injection, USP, 20 units/mL, Vial
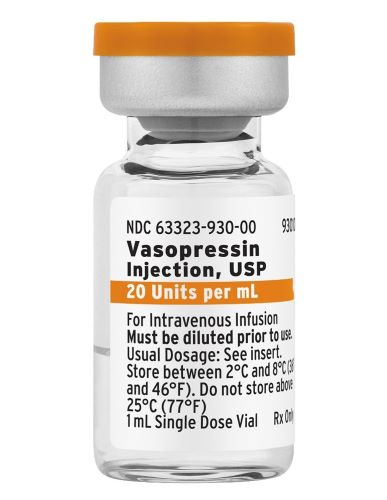
B. Image of US-Authorized Vasopressin Injection, USP, 20 units/mL, Inner Vial Label

|
NDC 63323-930-00 930101 Vasopressin For Intravenous Infusion |
Fresnius Kabi
|
C. Image of US-Authorized Vasopressin Injection, USP, 20 units/mL, Outer Carton Tray Label

|
NDC 63323-930-01 Rx Only 20 Units per mL For Intravenous Infusion Must be diluted prior to use |
Preservative Free |
What you should do
See Key Messages below
Additional information
Details
Get notified
Receive emails about new and updated recall and safety alerts.