COMIRNATY (COVID-19 Vaccine, mRNA, also referred to as Pfizer-BioNTech COVID-19 Vaccine): New Formulation/Presentation for Use in Individuals 12 Years of Age and Older
Summary
See Key Messages below
Affected products
COMIRNATY (COVID-19 Vaccine, mRNA, also referred to as Pfizer-BioNTech COVID-19 Vaccine) for 12 Years of Age and Older. Gray cap and gray label border - Do NOT dilute (30 mcg/0.3 mL). Suspension for intramuscular injection, multiple dose vials. Each vial contains 6* doses (each dose is 0.3 mL).
[DIN: 02522454]
Manufacturer: BioNTech Manufacturing GmbH (Germany)
Canadian Importer and Distributor: Pfizer Canada ULC
* Low dead-volume syringes and/or needles can be used to extract 6 doses from a single vial. If standard syringes and needles are used, there may not be sufficient volume to extract a 6th dose from a single vial.
Issue
UPDATED INFORMATION – June 10, 2022
Further to the communication below, issued on May 9, 2022, Health Canada has issued a new DIN (i.e. DIN 02527863) for COMIRNATY (COVID-19 Vaccine, mRNA), 30 mcg/0.3 mL, which has a GRAY vial cap and GRAY label border.
Each presentation of COMIRNATY in Canada now has a distinct DIN:
- GRAY CAP and GRAY LABEL BORDER (DIN 02527863)
- PURPLE CAP and PURPLE LABEL BORDER (DIN 02509210)
- ORANGE CAP and ORANGE LABEL BORDER (DIN 02522454).
The new DIN will help to mitigate potential medication errors between different presentations of COMIRNATY available in Canada. It will also facilitate tracking of product use by healthcare professionals.
May 9, 2022
COMIRNATY received a Notice of Compliance (NOC) under the Food and Drug Regulations on September 16, 2021, replacing the previous authorization under the Interim Order.
On March 15, 2022, COMIRNATY also received a NOC for an additional formulation/presentation (DIN 02522454) to be used in individuals 12 years of age and older. This new formulation/presentation does NOT require dilution.
In addition, as an extraordinary measure to provide access to vaccine supplies in the context of the global pandemic, Pfizer and BioNTech are continuing to provide vaccine supplies with vials and cartons labelled with the name Pfizer-BioNTech COVID-19 Vaccine. This label is presented in English-only and is missing some important Canadian-specific information normally found on Health Canada approved labels (see the “Information for healthcare professionals” section).
Audience
Healthcare professionals including infectious disease physicians, pharmacists, family physicians, public health officials, nurses and nurse practitioners. Healthcare professionals at the identified Points of Use.
Key messages
- On March 15, 2022, a new formulation/presentation of COMIRNATY (COVID-19 Vaccine, mRNA), 30 mcg/0.3 mL (DIN 02522454) for use in individuals 12 years of age and older, was authorized by Health Canada.
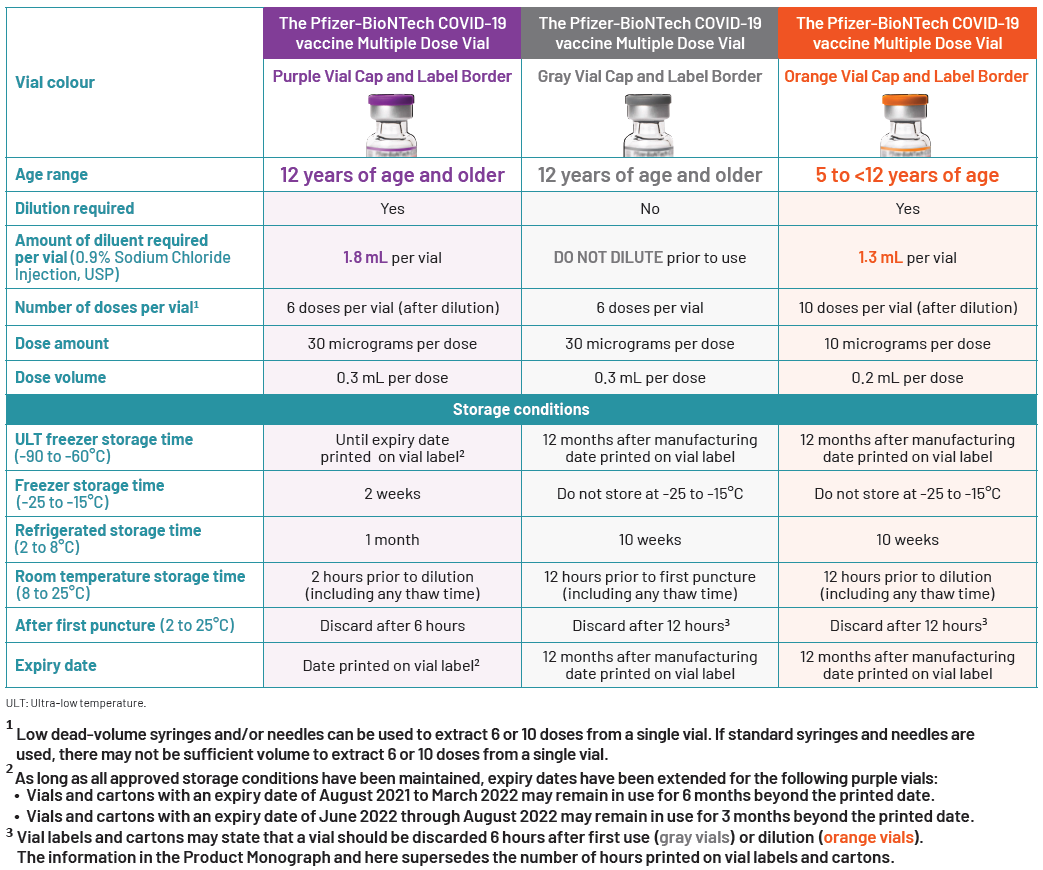
- This new formulation/presentation, which has a GRAY vial cap and GRAY label border, does NOT require dilution and has different storage requirements (see Table 1).
- COMIRNATY is now authorized as three different formulations /presentations, which are clearly differentiated by their vial cap and label border colours (see Appendices A, B and C):
- GRAY CAP AND GRAY LABEL BORDER
- 30 mcg/0.3 mL – Do NOT dilute
- For use in individuals 12 years of age and older
- PURPLE CAP AND PURPLE LABEL BORDER
- 30 mcg/0.3 mL after dilution
- For use in individuals 12 years of age and older
- ORANGE CAP AND ORANGE LABEL BORDER
- 10 mcg/0.2 mL after dilution
- For use in children 5 to less than 12 years of age
- GRAY CAP AND GRAY LABEL BORDER
- Each formulation/presentation has specific age authorizations, storage, handling, and preparation requirements. The differences are noted in Table 1 below, and it is important that this information be carefully reviewed prior to use. The formulation/presentation and lot number should be documented on patient vaccine records and for the purposes of adverse reaction reporting.
- When prepared according to their respective instructions, COMIRNATY with GRAY cap/label border and COMIRNATY with PURPLE cap/label border can be used interchangeably to provide the COVID-19 vaccination series for individuals 12 years of age and older.
- Vials of COMIRNATY intended for individuals 12 years of age and older (GRAY or PURPLE cap/label border) cannot be used to prepare doses for children 5 to less than 12 years of age.
- Although the vaccine’s brand name is now COMIRNATY, Canada will continue to receive vials of the vaccine labelled as Pfizer-BioNTech COVID-19 Vaccine. Pfizer Canada ULC is providing vaccine supplies with US Emergency Use Authorization English-only labels on the vials and cartons to expedite the global distribution of COMIRNATY.
- Healthcare professionals are advised that:
- Important Canadian-specific information is absent from the vial and carton labels (see the “Information for healthcare professionals” section).
- The Canadian-specific labelling information, including the COMIRNATY Product Monograph and training materials, can be accessed at CVDvaccine.ca or COMIRNATY.ca, or by scanning the QR code on the English-only carton label. This information is also available on the federal government’s covid-vaccine.canada.ca website. The COMIRNATY Canadian Product Monograph in French and English is also available on Health Canada’s Drug Product Database or at pfizer.ca.
Background
A new formulation/presentation of COMIRNATY 30 mcg/0.3 mL for use in individuals 12 years of age and older has been authorized by Health Canada. This new formulation/presentation does NOT require dilution, and has different storage requirements (see Table 1). It is packaged in vials with a gray cap and gray label border.
Given the public health emergency resulting from the current pandemic, Health Canada has authorized the importation, sale, and advertising of this new formulation/presentation with vial and carton labels that are in English-only for the current global distribution of the vaccine.
The Canadian Product Monograph for COMIRNATY, which is approved by Health Canada and available in French and English, should be used for complete product information.
Information for healthcare professionals
COMIRNATY is now authorized as three different formulations/presentations (see Appendices A, B and C):
- GRAY CAP AND GRAY LABEL BORDER
- 30 mcg/0.3 mL – Do NOT dilute
- For use in individuals 12 years of age and older
- PURPLE CAP AND PURPLE LABEL BORDER
- 30 mcg/0.3 mL after dilution
- For use in individuals 12 years of age and older
- ORANGE CAP AND ORANGE LABEL BORDER
- 10 mcg/0.2 mL after dilution
- For use in children 5 to less than 12 years of age
IMPORTANT: Each formulation/presentation has specific age authorizations, storage, handling, and preparation requirements. The differences are noted in Table 1 below, and it is important that this information be carefully reviewed prior to use. The formulation/presentation and lot number should be documented on patient vaccine records and for the purposes of adverse reaction reporting.
Vials of COMIRNATY with a gray cap and label border do NOT require dilution and are intended for use in individuals 12 years of age and older.
When prepared according to their respective instructions, COMIRNATY with gray cap/label border and COMIRNATY with purple cap/label border can be used interchangeably to provide the COVID-19 vaccination series for individuals 12 years of age and older.
Only vials of COMIRNATY with orange cap and label border are authorized to be used to prepare doses for children 5 to less than 12 years of age.
Table 1: Important Differences between the three COMIRNATY Formulations/Presentations (adapted from the COMIRNATY Product Monograph)
Healthcare professionals are advised that:
- Canadian-specific information can be accessed on CVDvaccine.ca or COMIRNATY.ca, or by scanning the QR code on the carton label. This information is also available on the federal government’s covid-vaccine.canada.ca website. The COMIRNATY Canadian Product Monograph in French and English is also available on Health Canada’s Drug Product Database or at pfizer.ca.
- The following important Canadian-specific information is absent from the vial and carton labels:
- Drug Identification Number (DIN)
- name and address of the Canadian DIN holder
- name and address of the Canadian importer and distributor
- all corresponding text in French
- The “COMIRNATY” brand name
- The vial and/or carton labels for the current supplies of vaccine include the statements “For use under Emergency Use Authorization.” The US Food and Drug Administration (FDA) specific information (e.g., Rx only, NDC) should be disregarded as this is not relevant to the Canadian authorization.
- For any medical questions, contact Pfizer Canada ULC Medical Information at 1-800-463-6001.
- For any other general inquiries, contact Pfizer Canada ULC Customer Service at 1-833-VAX-COVI (1-833-829-2684) or email at CanadaCSVaccine@pfizer.com
Action taken by Health Canada
Health Canada is permitting the use of an English-only label for a limited period. Health Canada has imposed terms and conditions requiring Pfizer Canada ULC to provide vaccine supplies with Canadian-specific labels as soon as feasible. Health Canada has made full labelling information available in French and English on the federal government’s covid-vaccine.canada.ca website.
Health Canada is communicating this important safety information to healthcare professionals and Canadians via the Recalls and Safety Alerts Database on the Healthy Canadians Web Site. This communication update will be further distributed through the MedEffect™ e-Notice email notification system, as well as through social media channels, including LinkedIn and Twitter.
Report health or safety concerns
Managing marketed health product-related side effects depends on healthcare professionals and consumers reporting them. Any serious or unexpected side effects in patients receiving COMIRNATY should be reported to your local Health Unit or Pfizer Canada ULC.
Pfizer Canada ULC
17300 Trans-Canada Highway
Kirkland, QC
H9J 2M5
Telephone: 1-866-723-7111
Fax: 1-855-242-5652
To correct your mailing address or fax number, contact Pfizer Canada ULC Customer Service at 1-833-VAX-COVI (1-833-829-2684).
If a patient experiences a side effect following immunization, please complete the Adverse Events Following Immunization (AEFI) Form appropriate for your province/territory and send it to your local Health Unit.
For other health product inquiries related to this communication, contact Health Canada at:
Biologic and Radiopharmaceutical Drugs Directorate
E-mail: brdd.dgo.enquiries@hc-sc.gc.ca
Original signed by
Vratislav Hadrava M.D., Ph.D.
Vice President & Medical Director
Pfizer Canada ULC
Reference
1. COMIRNATY (COVID-19 Vaccine, mRNA) [product monograph]. Mainz (Germany): BioNTech Manufacturing GmbH; 2022.
Appendix A
|
COMIRNATY (Pfizer-BioNTech COVID-19 Vaccine) for individuals 12 Years of Age and Older: Gray Cap and Label Border – Do NOT dilute
Vial and carton labels with English-only labelling |
VIAL (Gray Cap and Label Border)
CARTON (10-Vial)
Appendix B
|
COMIRNATY (Pfizer-BioNTech COVID-19 Vaccine) for individuals 12 Years of Age and Older: Purple Cap and Label Border - DILUTE BEFORE USE
Vial and carton labels with English-only labelling |
VIAL (Purple Cap and Label Border)
CARTON (195-Vial)
Appendix C
|
COMIRNATY (Pfizer-BioNTech COVID-19 Vaccine) for children 5 Years to <12 Years: Orange Cap and Label Border - DILUTE PRIOR TO USE
Vial and carton labels with English-only labelling |
VIAL (Orange Cap and Label Border)
CARTON (10-Vial)

Additional information
Details
Get notified
Receive emails about new and updated recall and safety alerts.