This page has been archived on the Web
Information identified as archived is provided for reference, research or recordkeeping purposes. It is not subject to the Government of Canada Web Standards and has not been altered or updated since it was archived. Please contact us to request a format other than those available.
Unauthorized OM Fusion Distributors LLC Health Products Found on the Canadian Market
- Starting date:
- February 19, 2010
- Posting date:
- February 19, 2010
- Type of communication:
- Advisory
- Subcategory:
- Natural health products
- Source of recall:
- Health Canada
- Issue:
- Important Safety Information, Unauthorized products
- Audience:
- General Public
- Identification number:
- RA-110004075
This document has been updated by an advisory dated June 3, 2010.
Health Canada is warning consumers of unauthorized health products sold under the OM Fusion Distributors LLC label included in the table below. The use of these products may cause life threatening reactions. There are serious risks with taking unauthorized drugs without supervision by a health care professional. If you take these products, you may also prevent your physician from properly diagnosing your condition and prescribing the correct medication.
Canadians who have purchased or used these products bearing the OM Fusion label are advised to immediately consult a health care practitioner. Consumers are advised to monitor the Health Canada Web site for additional updates regarding this issue.
The manufacturer packaging for the seven unauthorized products list certain ingredients that are drugs. These ingredients should only be sold with a prescription and are intended for use under the supervision of a health care professional only. It is illegal to sell to the public a product containing these ingredients unless the sale is made pursuant to a prescription.
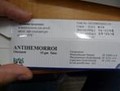
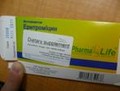
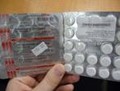
Each of these unauthorized pharmaceutical products have the same identifying image on their label (below) and have been sold without a prescription. The OM Fusion Distributors LLC label appears as a white sticker on top of the standard manufacturer packaging and falsely represents the prescription drugs as natural health products, such as skin care cream and dietary supplements.
The seven products detailed below are not authorized for sale in Canada and have been voluntarily forfeited to Health Canada by some retailers. These products were sold in Canada with manufacturer packaging that is labelled in only Russian, Ukrainian and Kazakh languages without English and/or French labelling. As a result, pertinent information about usage, dosage and side effects may not be available to the consumer.
| Product | Reported Drug Ingredient(s) |
|---|---|
| Adelphane - Esidrex | Hydrated Dihydralazine Sulphate |
| Antihemorroi Ointment | Betamethasone Valerate |
| Erythromycin | Erythromycin |
| Ftorocort | Triamcinolone Acetonide |
| Levomycetin | Chloramphenicol |
| Prednisolonum | Prednisolonum |
| Ampicillin - Zdorovye | Ampicillin |
Consumers should exercise caution when purchasing health products. Consumers are reminded that health products that are authorized for sale in Canada will have an eight-digit Drug Identification Number (DIN), a Natural Product Number (NPN) or a Homeopathic Medicine Number (DIN-HM) on the label. These numbers indicate that the products have been assessed by Health Canada for safety, effectiveness and quality.
Health Canada reminds retailers that the sale of unauthorized drugs contravenes the Food and Drugs Act. Sale of unauthorized drugs could render the seller liable for conviction for an offence under the Act. As such, Health Canada advises retailers to remove these products from their shelves and contact the Health Products and Food Branch Inspectorate for more information .
Health Canada also advises Canadians to contact the Health Products and Food Branch Inspectorate if they find these brands of products for sale in Canada. Complaints involving OM Fusion Distributors LLC can be reported to the Health Products and Food Branch Inspectorate by calling the toll-free hotline at 1-800-267-9675, or by writing to:
Health Products and Food Branch Inspectorate
Health Canada
Address Locator: 2003C
Ottawa, Ontario K1A 0K9
Or by E- mail: DCVIU_UVCEM@hc-sc.gc.ca
You can report any adverse reactions associated with the use of health products to the Canada Vigilance Program by one of the following three ways:
- Report online at the MedEffect™Canada section of the Health Canada website
- Call toll-free: 1-866-234-2345
-
Complete a Canada Vigilance Reporting Form and either:
- Fax toll-free: 1-866-678-6789
-
Mail to: Canada Vigilance Program
Health Canada
AL 0701D
Ottawa, ON K1A 0K9
To have postage pre-paid, download the postage paid label from the MedEffect™Canada Web site. The Canada Vigilance Reporting Form and the adverse reaction reporting guidelines may also be obtained via this Web site.
- Kazakh version (Download PDF document - 251 KB)
- Russian version (Download PDF document - 247 KB)
- Ukrainian version (Download PDF document - 254 KB)
Related AWRs
OM Fusion: Additional Unauthorized Products Removed from the Canadian Market
2010-06-03 | Health products
Media enquiries
Health Canada
613-957-2983
Public enquiries
613-957-2991
1-866-225-0709
Images
Select thumbnail to enlarge - opens in a new window