This page has been archived on the Web
Information identified as archived is provided for reference, research or recordkeeping purposes. It is not subject to the Government of Canada Web Standards and has not been altered or updated since it was archived. Please contact us to request a format other than those available.
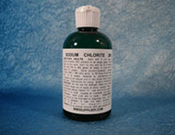
Sodium Chlorite Solution Not Authorized for Oral Consumption by Humans
- Starting date:
- February 15, 2012
- Posting date:
- February 15, 2012
- Type of communication:
- Advisory
- Subcategory:
- Drugs, Natural health products
- Source of recall:
- Health Canada
- Issue:
- Product Safety
- Audience:
- General Public
- Identification number:
- RA-110005307
The issue
Health Canada is advising Canadians that using "MMS", also known as Miracle Mineral Solution or Miracle Mineral Supplement may cause serious health problems. The product information lists sodium chlorite as an ingredient and is promoted as a substance that can cleanse toxins from the human body. There are no therapeutic products containing sodium chlorite authorized for oral consumption by humans in Canada.
These serious problems include poisoning, kidney failure and harm to red blood cells that reduces the ability of the blood to carry oxygen. Additional health problems may also include abdominal pain, nausea, vomiting, and diarrhoea.
Who is affected
Canadians who have purchased MMS associated with themmstore.
What consumers should do
- Consumers should consult their health care practitioner if they have used or are using MMS products.
- If consumers have questions such as disposal or refunds regarding MMS associated with themmstore, contact the distributor at 1-403-652-1655.
- Consumers should report any adverse reaction to Health Canada (see below).
What Health Canada is doing
Health Canada has notified the distributor associated with www.themsstore.com that the sale of sodium chlorite for human consumption is in contravention of the Food and Drugs Act. We have also requested that the distributor remove this product from the Canadian market, which they have agreed to do.
Health Canada has also identified and previously taken action on an unauthorized sodium chlorite solution. If additional products containing sodium chlorite promoted for oral consumption by humans are identified, Health Canada will add them to this list of affected products. Canadians are advised to monitor this list for any possible updates.
| Product Name | Ingredient | Retailer | Date Added |
|---|---|---|---|
|
Miracle Mineral Solution |
28% Sodium chlorite solution | Sold online: MMS Suppliers | February 8. 2012 |
|
MMS 1 |
28% Sodium chlorite solution | Sold online: The MMS Store | February 8, 2012 |
|
Miracle Mineral Supplement 2 |
27% and 28% Sodium chlorite solution | Sold online: Miracle Mineral Supplement | February 23, 2012 |
|
MMS 2 |
28% Sodium chlorite solution | Sold online: MMS1 | February 24, 2012 |
|
MMS 3 |
28% Sodium chlorite solution | Sold online: The MMS Store | May 18, 2012 |
Background
MMS distributed by www.themmstore.com claims to contain 28% sodium chlorite solution. Based on these claims, when taken as directed, MMS provides approximately 200 times more sodium chlorite than the tolerable daily intake of sodium chlorite in water established by Health Canada.
Reports to Health Canada indicate that ingestion of this product has been associated with four adverse reactions in Canada, including one life threatening reaction.
Sodium chlorite is authorized in Canada for use as a germicide by veterinarians and as a hard surface disinfectant.
For more information
Consumers and health professionals wanting more information about this advisory from Health Canada can contact the Public Enquiries Line at 613-957-2991, or toll free at 1-866-225-0709.
Media enquiries related to this Advisory should be directed to Health Canada Media Relations at 613-957-2983.
How to report side effects to health products
To report suspected adverse reaction to these or other health products, please contact the Canada Vigilance Program of Health Canada toll-free at 1-866-234-2345, or complete a Canada Vigilance Reporting Form and send to us using one of these methods:
Fax: 1-866-678-6789
E-mail: CanadaVigilance@hc-sc.gc.ca
Internet: MedEffect Canada
Mail:
Canada Vigilance Program
Marketed Health Products Directorate
Ottawa, ON, Address Locator 0701C
K1A 0K9
Related Health Canada Web Content
Related AWRs
Miracle Mineral Solution: Ingesting bleach-like chemical dangerous to health
2015-03-26 | Health products
Media enquiries
Health Canada
613-957-2983
Public enquiries
613-957-2991
1-866 225-0709
Images
Select thumbnail to enlarge - opens in a new window