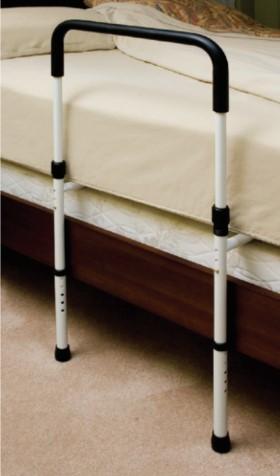
Essential Medical Supply - Endurance® Hand Bed Rails recalled due to entrapment and asphyxiation hazards
Brand(s)
Summary
Immediately stop using the recalled product and dispose of it.
Affected products
This recall involves certain Essential Medical Supply - Endurance® Hand Bed Rails manufactured between October 2006 and December 2021.
| Product | Model Number |
|---|---|
| Hand Bed Rail | P1410 |
| Hand Bed Rail with Pouch | P1410-P |
| Hand Bed Rail with Floor Support | P1411 |
| Hand Bed Rail with Floor Support with Pouch | P1411-P |
Issue
When attached to an adult’s bed, users can become entrapped within the bed rail or between the rail and the side of the mattress. This poses a serious entrapment hazard and risk of death by asphyxiation.
As of April 2024, the company has received no reports of incidents in Canada, and no reports of injuries. In the United States, the company has received 3 reports of incidents and 4 reported deaths.
What you should do
Consumers should immediately stop using the recalled Endurance® Hand Bed Rails and dispose of them.
For more information, consumers can contact Essential Medical Supply by telephone at 800-826-8423 from 8am to 5pm ET, Monday through Friday or online at www.essentialmedicalsupply.com.
Please note that the Canada Consumer Product Safety Act prohibits recalled products from being redistributed, sold or even given away in Canada.
Health Canada would like to remind consumers to report any health or safety incidents related to the use of this product or any other consumer product or cosmetic by filling out the Consumer Product Incident Report Form.
This recall is also posted on the OECD Global Portal on Product Recalls website. You can visit this site for more information on other international consumer product recalls.
Additional information
Background
Number Sold
The company reported that 20 units of the affected products were sold in Canada and 272,000 were sold in the United States.
Time Period Sold
The affected products were sold from October, 2006 to December, 2021.
Place of Origin
Manufactured in Taiwan
Details
Distributor
Essential Medical Supply
Florida
United States
Get notified
Receive emails about new and updated recall and safety alerts.