This page has been archived on the Web
Information identified as archived is provided for reference, research or recordkeeping purposes. It is not subject to the Government of Canada Web Standards and has not been altered or updated since it was archived. Please contact us to request a format other than those available.
Additional unauthorized health products labelled as B17/amygdalin/bitter apricot kernel may pose serious risk to health
- Starting date:
- February 25, 2016
- Posting date:
- February 25, 2016
- Type of communication:
- Advisory
- Subcategory:
- Natural health products
- Source of recall:
- Health Canada
- Issue:
- Poisoning Hazard
- Audience:
- General Public
- Identification number:
- RA-57272
Issue
Health Canada is advising Canadians that a number of potentially dangerous unauthorized health products labelled as B17/amygdalin/bitter apricot kernel (see below) were available for sale in Canada. Further to Health Canada’s previous communication, no health products containing B17 or amygdalin have been authorized to treat cancer or any other condition. Patients who have purchased any of these products to treat cancer should stop using the product and contact their doctor for appropriate follow-up.
Products containing apricot kernel extract may contain amygdalin, a compound derived from bitter apricot kernels that has the potential to release cyanide when ingested by humans. The concentration of amygdalin in the products identified below is not known at this time. Ingesting low to moderate amounts of cyanide may lead to serious adverse health consequences and high doses may be lethal.
Products affected:
| Photo | Product | Indicated use | Seller | Web site | Date added |
|---|---|---|---|---|---|
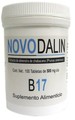
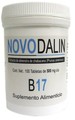
| Novodalin B17 | Cancer | Discount Natural Foods | CDNF.com | 23-Feb-2016 | |
| Novodalin B17 | Dietary supplement | Discount Natural Foods | Amazon.ca | 25-Feb-2016 | |
| B17 Amygdalin | Dietary supplement | Discount Natural Foods | Amazon.ca | 25-Feb-2016 | |
| Body Renew 12 in 1 Formula | Dietary supplement | Discount Natural Foods | Amazon.ca | 25-Feb-2016 | |
| Body Me Organic Bitter Apricot Kernel | Dietary supplement | Beauty Outlet | Amazon.ca | 25-Feb-2016 |
Who is affected:
Consumers who have bought or used the affected products.
What consumers should do
- Consult with your doctor if you have used these products and have health concerns.
- Read the product label to verify that health products have been authorized for sale by Health Canada have an eight-digit DIN, a DIN-HM or a NPN.
- Verify if products that have been authorized for sale by searching the Drug Product Database and the Licensed Natural Health Product Database.
- Report any adverse events to Health Canada.
- Report complaints about health products to Health Canada by calling toll-free to 1-800-267-9675, or complete an online complaint form.
What Health Canada is doing:
Health Canada has requested that the products be removed from sale and will take further action and advise Canadians should additional risks be identified.
Background:
Health Canada has previously communicated on the risk of buying drugs, natural health products or medical devices online. The healthycanadians website also routinely informs consumers about unlicenced health products that have been found on the Canadian market.
Unlicensed products have not been reviewed by Health Canada for safety, quality or effectiveness. They may contain hidden ingredients not listed on the label, dangerous additives and/or contaminated ingredients. In addition, they may lack the active ingredients Canadians would expect them to contain to help maintain and improve their health. For all of these reasons, they could cause serious health effects.
Report health or safety concerns
How to report side effects to health products to Health Canada:
- Call toll-free at 1-866-234-2345
- Visit MedEffect Canada's web page on Adverse Reaction Reporting for information on how to report online, by mail or by fax.
Stay connected with Health Canada and receive the latest advisories and product recalls using social media tools.
Media enquiries
Health Canada
(613) 957-2983
Public enquiries
(613) 957-2991
1-866 225-0709
Images
Select thumbnail to enlarge - opens in a new window